An article written by Dr Edward Leatham, Consultant Cardiologist © 2026 E.Leatham
For busy people, or to tune in when on the move, Google Notebook AI audio podcast and an explainer slide show are available for this story beneath.
Statins, Absolute Benefit, and the Problem of Residual Risk
Statins have transformed cardiovascular prevention. Few drug classes have such a consistent and robust evidence base demonstrating reductions in myocardial infarction, stroke, and cardiovascular mortality. Lowering low-density lipoprotein cholesterol (LDL-C) clearly reduces atherosclerotic risk, and statins remain foundational therapy in both primary and secondary prevention.
However, when we move beyond headlines and examine absolute benefit, a more uncomfortable reality emerges. Even with optimal LDL-C lowering over the typical duration of modern cardiovascular trials — usually 2 to 3 years — the majority of cardiovascular events are not prevented. This ongoing burden is referred to as residual cardiovascular risk, and it represents one of the most important unmet needs in contemporary preventive cardiology.
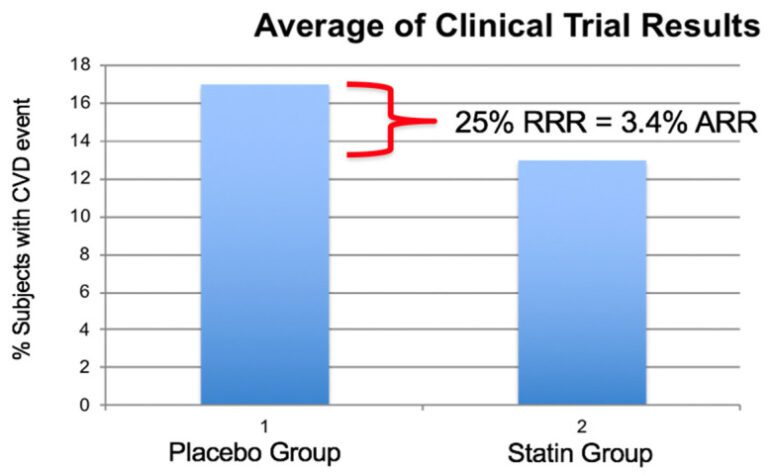
Percent of subjects experiencing a cardiovascular event in eight large statin investigations Scandinavian Simvastatin Survival Study (SSSS), Pravastatin or Atorvastatin Evaluation and Infection Therapy–Thrombolysis in Myocardial Infarction 22 (PROVE-IT 22), Heart Protection Study (HPS), Long-Term Intervention with Pravastatin in Ischaemic Disease (LIPID), Cholesterol and Recurrent Events (CARE), Treating to New Targets (TNT), Air Force/Texas Coronary Atherosclerosis Prevention Study (AFTEXCAPS), West of Scotland Coronary Prevention Study (WOSCOPS).
Average relative risk reduction was 25% and the absolute risk reduction was 3.4% illustrating the large number of subjects experiencing a cardiovascular (CV) event while on statin therapy with reduced LDL-C values. (Modified from reference (1)).

Figure reproduced unmodified from: Superko HR, Garrett B. Small dense LDL: scientific background, clinical relevance, and recent evidence still a risk even with “normal” LDL-C levels. Biomedicines. 2022;10(4):829. © 2022 The Authors. Distributed under the Creative Commons Attribution (CC BY 4.0) licence.
An important factor underpinning this observation is the design of modern clinical trials. Due to the substantial cost of running large cardiovascular outcome studies, most commercially funded trials enrol large numbers of patients to ensure statistical power, but observe them over relatively short periods. While this approach is sufficient to demonstrate biological efficacy, it inevitably underestimates the full lifetime benefit of therapy. The assumption is that a treatment effect observed at 2–3 years will continue — and likely widen — over longer horizons of 5, 10, or even 20 years. Such durations, however, are impractical for randomised trials, leaving long-term benefit inferred rather than proven. This limitation reinforces a core principle of preventive cardiology: earlier intervention in at-risk populations is likely to yield the greatest cumulative benefit, even if short-term absolute risk reductions appear modest.
The importance of residual risk lies not in diminishing the value of statins, but in recognising that other pathogenic factors, beyond LDL-C, are also at play. Atherosclerosis is biologically complex, and lowering LDL-C addresses only one component of risk. This recognition has, at times, led some critics to question whether the benefits of statins arise primarily from LDL-C reduction or from so-called pleiotropic effects, such as anti-inflammatory or endothelial actions.
However, this argument has been substantially weakened by consistent evidence showing that cardiovascular risk reduction closely tracks the degree of LDL-C lowering, regardless of how that lowering is achieved. Whether LDL-C is reduced by statins, ezetimibe, or PCSK9 inhibitors, the magnitude of clinical benefit is proportional to the absolute reduction in LDL-C. This concordance strongly reinforces the conclusion that the principal driver of statin benefit is LDL-C lowering itself, rather than off-target or ancillary effects.
Residual risk therefore does not negate the LDL hypothesis. Instead, it highlights that LDL-C lowering is necessary but not sufficient to fully address cardiovascular risk.
Relative Risk vs Absolute Risk: Why the Difference Matters
Statin trials are often presented in terms of relative risk reduction (RRR), typically around 20–30%. While impressive, relative risk can obscure how much risk is actually removed for an individual patient.
When outcomes are expressed as absolute risk reduction (ARR) and Number Needed to Treat (NNT), the scale of benefit — and the scale of what remains — becomes clearer.

In JUPITER, a primary prevention trial in patients with normal LDL-C but elevated hs-CRP, 83 patients needed to be treated for just under two years to prevent one major cardiovascular event(2). In FOURIER, despite background statin therapy and LDL-C driven down to ~30 mg/dL using a PCSK9 inhibitor, around 67 patients required treatment for ~2.5 years to prevent one event(3).
These are clinically meaningful benefits — but they also demonstrate that the majority of treated patients remain at risk.
Residual Risk Persists Even with Very Low LDL-C
The FOURIER trial was particularly revealing. LDL-C levels previously considered “ideal” — and then some — did not abolish cardiovascular events. Event curves continued to diverge slowly, but they never flattened. This observation forces an important conclusion:
LDL-C is necessary to treat, but insufficient to explain total cardiovascular risk.
Superko and Garrett highlight a key contributor to this residual risk: lipoprotein heterogeneity, particularly small, dense LDL (sdLDL) (1).
Small, Dense LDL: Risk Beyond LDL-C
Not all LDL particles behave the same. sdLDL particles are:
- More likely to penetrate the arterial wall
- Less readily cleared from circulation
- More susceptible to oxidative modification
Multiple studies show that sdLDL is associated with coronary heart disease independent of LDL-C levels, including in patients whose LDL-C is “normal” or well-controlled on statins(1,4,5).

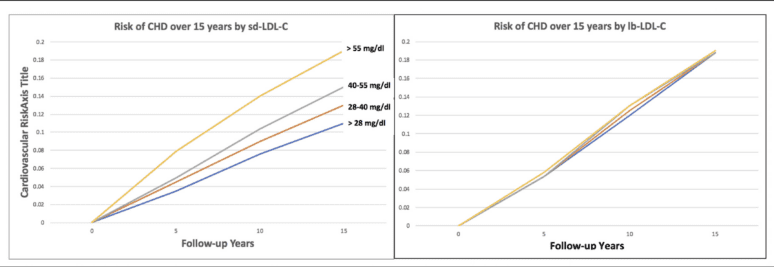
Small dense LDL-C (sd-LDL-C) quartiles and large buoyant LDL-C (lb-LDL-C) quartiles and cardiovascular risk over 15 years of follow-up in ARIC. (Modified from reference(1,6)).
Figure reproduced unmodified from: Superko HR, Garrett B. Small dense LDL: scientific background, clinical relevance, and recent evidence still a risk even with “normal” LDL-C levels. Biomedicines. 2022;10(4):829. © 2022 The Authors. Distributed under the Creative Commons Attribution (CC BY 4.0) licence.
This is residual risk, quantified.
Statins reduce LDL-C concentration, but they do not reliably normalise LDL particle size or number, particularly in patients with underlying metabolic dysfunction. This raises the question: what drives sdLDL in the first place?
Insulin Resistance and Visceral Adipose Tissue: The Metabolic Engine of Residual Risk
A major, and often under-recognised, driver of residual cardiovascular risk is insulin resistance, closely linked to excess visceral adipose tissue (VAT).
Visceral fat is not passive energy storage. It is metabolically active and releases free fatty acids directly into the portal circulation, driving hepatic overproduction of VLDL. This results in the classic atherogenic dyslipidaemia pattern:
- Elevated triglycerides
- Low HDL-cholesterol
- Increased sdLDL particles
This phenotype is strongly associated with insulin resistance and metabolic syndrome(4,7,8).
Crucially, statins only partially address this lipid pattern. While LDL-C falls, triglyceride-rich lipoproteins, sdLDL, inflammation, and endothelial dysfunction often persist.
In practical terms, this means the biological drivers of atherosclerosis remain active, even when LDL-C looks excellent on a lipid panel.
Why This Explains Residual Risk in Statin and PCSK9 Trials
Residual risk persists because:
- LDL-C lowering does not fully address particle number, particle quality, or triglyceride-rich lipoproteins
- Insulin resistance and VAT promote inflammation, oxidative stress, and thrombogenicity
- Atherosclerosis is metabolically driven, not cholesterol-driven alone
This explains why:
- Event rates remain substantial in statin-treated populations
- Adding PCSK9 inhibition lowers risk further, but modestly in absolute terms
- Cardiovascular disease continues to develop in patients with “optimal” LDL-C
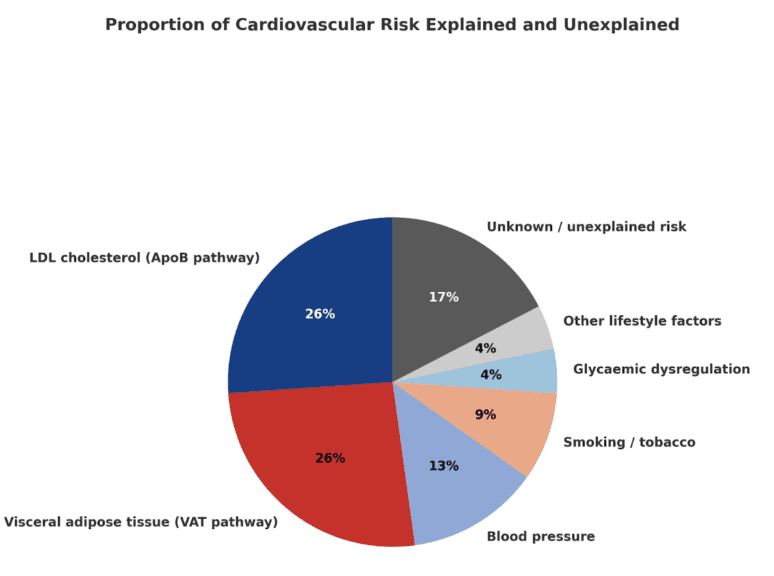
How to Interpret These Percentages

Each slice of the chart shows the share of total heart disease in the population attributed to that factor — not the proportion of a single person’s risk.
For example:
- Smoking ≈ 10 % means about 1 in 10 heart attacks in the UK are caused by smoking.
Yet an individual smoker’s risk of heart disease is roughly double that of a non-smoker. - LDL cholesterol (≈ 30 %) means about one-third of heart disease cases could be prevented if everyone achieved optimal LDL levels.
- Visceral fat (≈ 30 %) means around one-third is linked to excess visceral or ectopic fat.
These figures describe population impact, not how the risks divide within one person.
They help guide priorities in prevention — targeting LDL, visceral fat, blood pressure, and smoking yields the largest health gains for the community.
Implications for Prevention
Statins remain essential. They reduce risk and save lives. But they should be viewed as foundation therapy, not the endpoint.
Meaningful reduction in residual risk requires:
- Attention to triglycerides, sdLDL, apoB, and non-HDL cholesterol
- Targeting metabolic health alongside lipid lowering
- Integrating lifestyle, metabolic, and — where appropriate — adjunctive pharmacological strategies
Only by addressing both LDL-mediated risk and VAT-driven metabolic risk can we realistically hope to shrink the large residual burden of cardiovascular disease that persists despite modern lipid therapy.
Bibliography
- Relative Contributions Of LDL VAT And Other Cardiovascular Risk Factors In The UK 2025 by Dr Edward Leatham
- Bradford Hill Appraisal of Raised Visceral Adipose Tissue causing Coronary Heart Disease 2025 Dr Edward Leatham
References
- Superko H, Garrett B. Small Dense LDL: Scientific Background, Clinical Relevance, and Recent Evidence Still a Risk Even with ‘Normal’ LDL-C Levels. Biomedicines [Internet]. 2022 Apr 1 [cited 2025 Dec 15];10(4):829. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC9025822/
- Ridker PM, Danielson E, Fonseca FAH, Genest J, Gotto AM, Kastelein JJP, et al. Rosuvastatin to Prevent Vascular Events in Men and Women with Elevated C-Reactive Protein. N Engl J Med [Internet]. 2008 Nov 20 [cited 2025 Dec 4];359(21):2195–207. Available from: https://www.nejm.org/doi/full/10.1056/NEJMoa0807646
- Sabatine MS, Giugliano RP, Keech AC, Honarpour N, Wiviott SD, Murphy SA, et al. Evolocumab and Clinical Outcomes in Patients with Cardiovascular Disease. N Engl J Med [Internet]. 2017 May 4 [cited 2025 Dec 4];376(18):1713–22. Available from: https://www.nejm.org/doi/full/10.1056/NEJMoa1615664
- Krauss RM. Small dense low-density lipoprotein particles: clinically relevant? Curr Opin Lipidol [Internet]. 2022 Jun 1 [cited 2025 Dec 15];33(3):160–6. Available from: https://escholarship.org/uc/item/9xm7v937
- Austin MA, King MC, Vranizan KM, Krauss RM. Atherogenic lipoprotein phenotype. A proposed genetic marker for coronary heart disease risk. Circulation. 1990 Aug;82(2):495–506.
- Hoogeveen RC, Gaubatz JW, Sun W, Dodge RC, Crosby JR, Jiang J, et al. Small Dense Low-Density Lipoprotein-Cholesterol Concentrations Predict Risk for Coronary Heart Disease. Arterioscler Thromb Vasc Biol [Internet]. 2014 May [cited 2026 Feb 9];34(5):1069–77. Available from: https://www.ahajournals.org/doi/10.1161/ATVBAHA.114.303284
- Després JP. Body Fat Distribution and Risk of Cardiovascular Disease. Circulation [Internet]. 2012 Sep 4 [cited 2025 Dec 13];126(10):1301–13. Available from: https://www.ahajournals.org/doi/10.1161/circulationaha.111.067264
8. Adiels M, Olofsson SO, Taskinen MR, Borén J. Overproduction of very low-density lipoproteins is the hallmark of the dyslipidemia in the metabolic syndrome. Arterioscler Thromb Vasc Biol. 2008 Jul;28(7):1225–36.