An article written by Dr Edward Leatham, Consultant Cardiologist © 2026 E.Leatham
For busy people, or to tune in when on the move, Google Notebook AI audio podcast are available for this story beneath.
What a hybrid car can teach us about cardiac metabolism
If you wanted to design the perfect engine, you would probably start with something highly efficient, endlessly adaptable, and capable of switching fuels depending on demand. You might imagine a sophisticated hybrid vehicle — one that uses electric power for rapid acceleration, fuel combustion for long-distance efficiency, and intelligent systems to optimise performance under changing conditions.
Nature has already built something better.
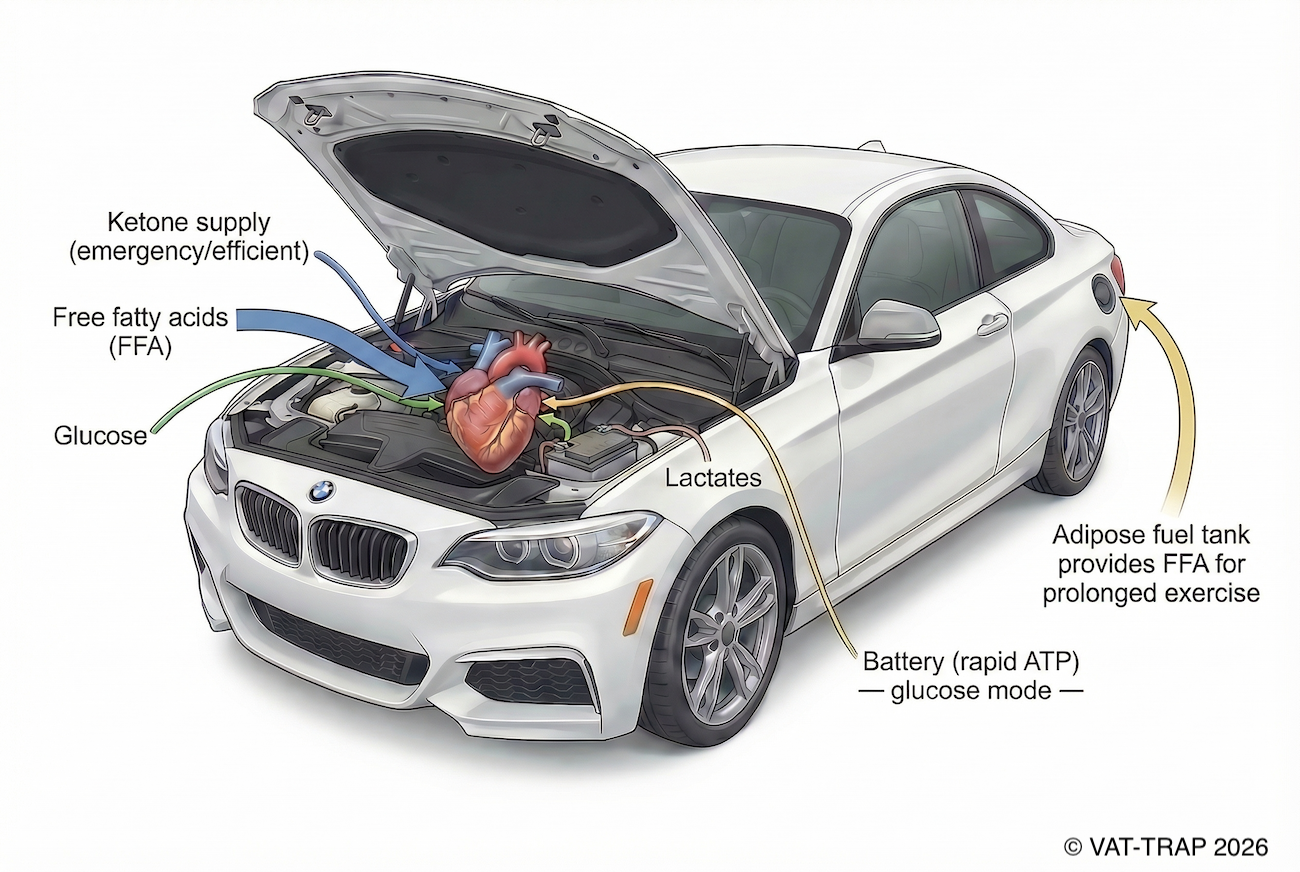
The human heart is the ultimate hybrid engine — a metabolic omnivore that continuously selects the best available fuel to keep us alive. Unlike any machine humans have designed, it does not rely on a single energy source. Instead, it flexibly switches between free fatty acids, glucose, and ketones, adjusting moment by moment to workload, oxygen availability, hormones, and nutritional state.
In many ways, the heart behaves like an advanced hybrid car — but with capabilities that would make any engineer jealous.
A pump that never stops
Before exploring fuels, it is worth remembering the scale of the task.
The heart beats roughly:
- 100,000 times per day
- Over 3 billion times in a lifetime
It must produce ATP continuously, with almost no rest periods. Energy failure is not an option.
Unlike skeletal muscle, the heart cannot pause and repay energy debt later. That means its energy system has evolved around two key principles:
- Efficiency
- Fuel flexibility
This is where the hybrid analogy becomes useful.
The hybrid model: electric vs diesel
Imagine a modern hybrid vehicle.
- The electric motor provides immediate torque, smooth acceleration, and efficient performance during high-demand moments.
- The diesel engine (or fuel engine) provides steady, long-range power and endurance.
The heart behaves in a similar way.
The electric mode — glucose
Glucose is like the electric motor:
- Rapidly available
- Oxygen-efficient per ATP generated
- Ideal for periods of stress or sudden increased demand
When the heart needs to work harder — during exercise, stress, or increased sympathetic drive — glucose metabolism becomes more prominent.
Why?
Because glucose oxidation produces more ATP per molecule of oxygen than fatty acids. In a situation where oxygen delivery becomes limiting, glucose offers a metabolic advantage.
In short:
Glucose is the heart’s “performance mode.”
The diesel mode — free fatty acids (FFA)
Most of the time, however, the heart runs on something else.
At rest in healthy adults, approximately 60–80% of myocardial energy comes from free fatty acids.
Fatty acids resemble diesel fuel:
- Highly energy dense
- Excellent for sustained output
- Ideal for continuous aerobic burning
They provide massive ATP yield and support the heart’s uninterrupted workload across decades.
From an engineering perspective, this is logical:
- The heart works continuously but usually at moderate intensity.
- A stable, high-energy fuel makes sense for baseline operation.
So under normal everyday conditions:
The heart cruises on fatty acids.
The heart beats the hybrid engine
The analogy becomes even more interesting because the heart goes beyond hybrid design.
A car has two modes — electric and fuel combustion.
The heart has more.
It can also run on:
- Ketone bodies
- Lactate
- Amino acids (to a limited extent)
This makes the heart less like a hybrid and more like an intelligent multi-fuel engine.
Ketones — the emergency reserve (or efficiency fuel?)
During fasting, prolonged exercise, or carbohydrate restriction, ketone bodies rise in the bloodstream.
The heart readily oxidises ketones and may even prefer them under certain conditions.
Ketones are:
- Efficient mitochondrial substrates
- Rapidly oxidised
- Oxygen-efficient relative to fatty acids
Some research suggests that the failing heart increases ketone uptake — possibly because ketones offer a metabolically favourable fuel when energy efficiency becomes compromised.
In other words:
The heart can quietly switch to a third energy mode when conditions change.
No vehicle on the road today can do this.
Why fuel flexibility matters
The real brilliance of cardiac metabolism is not any single fuel — it is the ability to switch rapidly between them.
This flexibility allows the heart to:
- Maintain constant ATP levels
- Adapt to changing nutrient availability
- Match oxygen demand and supply
- Survive periods of fasting or stress
A rigid engine would fail quickly in a variable environment.
The heart thrives because it is metabolically adaptive.

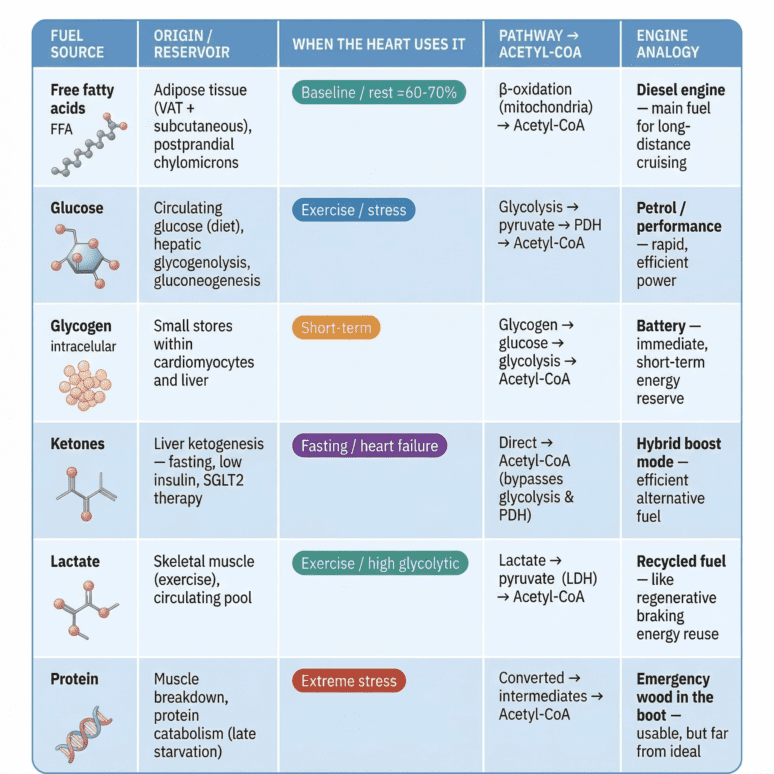
This table shows how the heart uses multiple fuels, each drawn from different body stores, much like a hybrid car switching energy sources. Fatty acids provide steady baseline power, glucose supports high demand, glycogen offers rapid reserve, and ketones act as an efficient alternative—highlighting the heart’s key strength: metabolic flexibility.
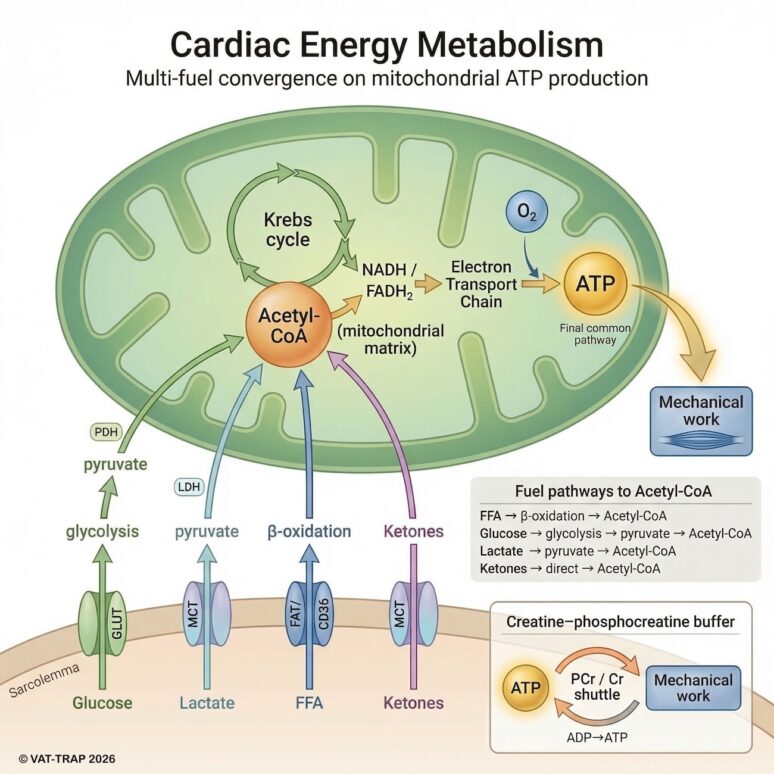
The metabolic conductor: mitochondria

At the centre of this flexibility are the mitochondria — the tiny power plants inside cardiac muscle cells.
The heart contains more mitochondria than almost any other tissue:
- Up to 30–40% of myocardial volume is mitochondrial.
These organelles constantly integrate:
- Hormonal signals (insulin, catecholamines)
- Substrate availability
- Oxygen tension
- Workload
The result is a continuously optimised fuel mix — like an onboard computer adjusting engine performance every second.
When the system begins to change
In metabolic disease, this balance begins to shift.
In insulin resistance and high visceral adiposity:
- Fatty acid delivery to the heart increases
- Glucose utilisation becomes impaired
- Metabolic flexibility declines
The heart becomes more reliant on fatty acid oxidation.
While fatty acids provide abundant energy, they are less oxygen-efficient, and excessive reliance may increase oxidative stress and mitochondrial dysfunction.
But the problem goes beyond fuel.
🛣️ The fuel lines: coronary arteries
Visceral adiposity and insulin resistance also affect the delivery system:
- Accelerated coronary atherosclerosis
- Endothelial dysfunction
- Microvascular impairment
In effect:
The fuel lines narrow while the engine becomes less efficient.
🔥 The fuel quality: inflammation
Visceral adipose tissue is not just a storage depot — it is metabolically active.
It releases:
- Pro-inflammatory cytokines (e.g. IL-6, TNF-α)
- Adipokines
- Oxidative mediators
This creates a state of chronic low-grade inflammation.
From an engine perspective:
It is like running the engine on contaminated fuel.
Consequences include:
- Mitochondrial dysfunction
- Impaired ATP production
- Increased oxidative stress
- Endothelial injury
So not only is the fuel mix suboptimal — the quality of the fuel is degraded.
⚙️ The pressure load: hypertension
High blood pressure adds a further layer.
Increased afterload means:
- The heart must generate higher الضغط to eject blood
- Wall stress increases
- Myocardial oxygen demand rises
In engine terms:
The engine is working against higher resistance — like driving uphill constantly.
Or more mechanically:
Increased pressure load accelerates wear and tear on the system.
Over time this leads to:
- Left ventricular hypertrophy
- Reduced efficiency
- Increased risk of failure
Like a car engine under constant strain, the risk of “mechanical failure” increases — the biological equivalent being myocardial dysfunction and heart failure.
🧠 Bringing it together
In cardiometabolic disease, three things go wrong simultaneously:
1️⃣ Fuel problem
- Excess fatty acids
- Impaired glucose use
- Reduced flexibility
2️⃣ Fuel quality problem
- Chronic inflammation
- Oxidative stress
3️⃣ Fuel delivery + load problem
- Coronary disease (narrowed supply lines)
- Hypertension (increased workload)
⭐ The key insight
In VAT-driven metabolic disease, the heart is forced to run on poorer-quality fuel, through narrowed supply lines, while working against higher pressure — a perfect setup for energy inefficiency and eventual failure.
In metabolic disease, this balance can shift.
In insulin resistance and high visceral adiposity:
- Fatty acid delivery to the heart increases.
- Glucose utilisation declines.
- Metabolic flexibility decreases.
The heart becomes more “locked” into fatty acid use.
While FFA provide energy, they are less oxygen-efficient, and excessive reliance can increase oxidative stress.
This helps explain why cardiometabolic disease is not only about vessels — it also affects myocardial metabolism itself.
The heart in heart failure: a fascinating paradox
One of the most interesting observations in modern cardiology is that failing hearts often shift fuel preference again.
Rather than relying predominantly on fatty acids, the failing heart may:
- Increase glucose uptake
- Increase ketone use
This appears to be an adaptive attempt to improve energetic efficiency when ATP production becomes threatened.
It is almost as if the engine senses fuel inefficiency and switches to a more economical mode.
Lessons from the hybrid analogy
The comparison to a hybrid car helps illustrate several key principles:
1. No single fuel is “good” or “bad”
The heart needs multiple fuels.
Problems arise not from fuel type alone but from loss of flexibility.
2. Efficiency changes with circumstances
- Glucose = high-performance, oxygen-efficient mode
- Fatty acids = long-distance endurance mode
- Ketones = adaptive backup or efficiency booster
3. Metabolic health means adaptability
A healthy heart switches fuels effortlessly.
A metabolically unhealthy heart becomes rigid — and less efficient.
Why this matters for the VAT Trap
High visceral adipose tissue changes fuel availability at the system level.
VAT drives:
- Increased free fatty acid flux
- Hepatic VLDL production
- Insulin resistance
This influences not only lipid profiles but also myocardial fuel exposure.
In many high-VAT individuals:
- Fatty acids dominate energy supply.
- Glucose utilisation becomes impaired.
- Metabolic flexibility is reduced.
Understanding this helps explain why improving metabolic health — through weight reduction, exercise, and therapies that improve insulin sensitivity — can have profound effects beyond cholesterol numbers alone.
Exercise: the ultimate fuel trainer
Exercise is perhaps the best way to restore fuel flexibility.
Training improves:
- Mitochondrial density
- Fat oxidation efficiency
- Glucose uptake
- Metabolic switching capacity
In hybrid terms:
Exercise upgrades the operating software of the engine.
The elegance of evolution
From an evolutionary perspective, this system makes perfect sense.
Humans historically experienced:
- Periods of feast and famine
- Variable activity levels
- Fluctuating food availability
A heart capable of switching between fuels would have a major survival advantage.
Modern life — constant caloric availability and sedentary behaviour — challenges this system in ways it was not designed for.
The bigger picture
When we think about cardiovascular risk, we often focus on:
- LDL cholesterol
- Blood pressure
- Weight
But beneath these markers lies a deeper truth:
The heart is a metabolic organ.
Its function depends not only on blood supply but also on fuel availability and flexibility.
Final thoughts: the engine that never stops
If a hybrid car represents the pinnacle of modern engineering, the human heart is something even more remarkable.
- It runs primarily on fatty acids for endurance.
- It shifts toward glucose for performance and efficiency.
- It seamlessly incorporates ketones when needed.
- It adapts continuously without conscious control.
And unlike any machine we have built, it never shuts down for maintenance.
Perhaps the best way to think of the heart is this:
A self-optimising, multi-fuel engine designed for lifelong performance — provided we give it the metabolic environment it evolved for.
One-line takeaway
The heart is the ultimate fuel omnivore — a biological hybrid engine that usually cruises on fatty acids, switches to glucose for peak efficiency, and can even run on ketones, illustrating the extraordinary metabolic flexibility that underpins cardiovascular health.
Related posts and downloads
Prediabetes as a Therapeutic Target: A Cardiologist’s Editorial Perspective
Unravelling the Conundrum of Glucose Spikes
From The Naked Heart to The VAT Trap
Fatty Liver
Could Reducing Belly Fat Improve Erectile Function?
Can We Use AI to Predict Cardiovascular Death?
Your Roadmap to CHD Prevention: A 4-Step Guide
The Three Most Preventable Causes of a Heart Attack
The Hidden Culprit Behind Heart Disease: Small Dense LDL and the Fat You Can’t See
LDL-C, Apo B and sdLDL targets: a guide for cardiologists and their patients