An article by Dr Edward Leatham, Consultant Cardiologist © 2026 E.Leatham
For busy people, or to tune in when on the move, Google Notebook AI audio podcast are available for this story beneath.
TOE Before Cardioversion: Essential Stroke Prevention in Atrial Fibrillation Transoesophageal echocardiography identifies hidden clots before cardioversion, preventing stroke from atrial paresis when normal rhythm returns.
A transoesophageal echocardiogram (TOE) is a test where a small ultrasound probe is gently passed into the oesophagus (food pipe) to take detailed pictures of the heart from behind. This allows us to clearly see areas such as the left atrial appendage, where blood clots can sometimes form.
In people with atrial fibrillation or atrial flutter, a clot may be present before restoring a normal heart rhythm with cardioversion. If a clot is dislodged, it can lead to a stroke. A TOE is therefore used to check for clots if adequate anticoagulation has not already been given.
If no clot is seen, cardioversion can usually proceed safely with continued blood-thinning medication. However, in this video, a clot is visible in the left atrial appendage. This means cardioversion must be delayed, and further anticoagulation is needed before reassessment.
When cardioverting atrial fibrillation, the greatest stroke risk comes not from existing clots, but from atrial paresis following rhythm restoration. TOE remains crucial for detecting left atrial appendage thrombus that transthoracic echo cannot visualise reliably.
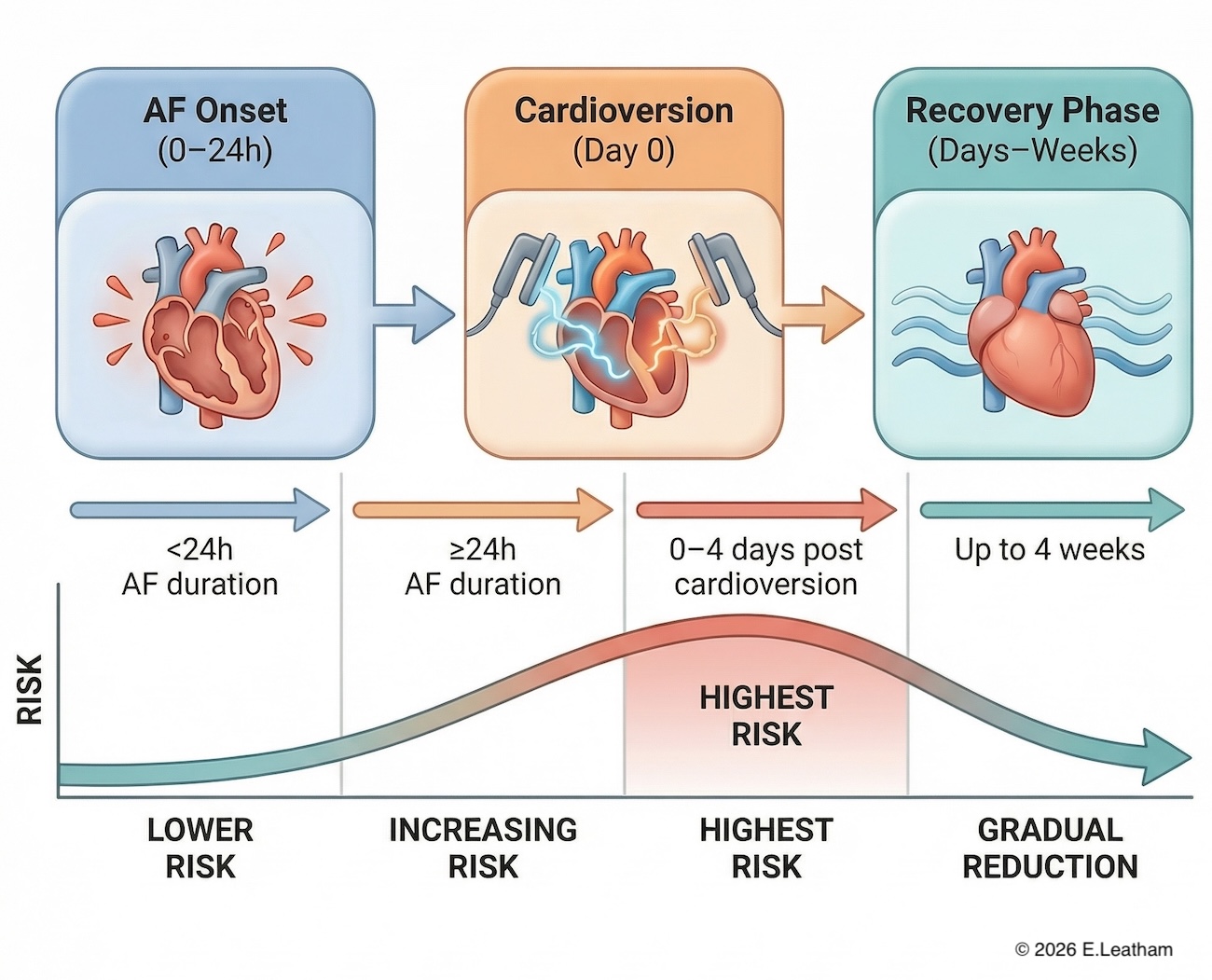
Background Atrial fibrillation affects over 1.4 million people in the UK, with cardioversion offering rapid symptom relief and rhythm restoration. However, the procedure carries significant stroke risk, particularly in the hours following successful conversion to sinus rhythm.
The left atrial appendage—a finger-like pouch extending from the left atrium—represents the primary site of thrombus formation in atrial fibrillation. This anatomical structure, with its complex trabeculated interior, creates perfect conditions for blood stasis and clot development when atrial contraction becomes chaotic.
Standard transthoracic echocardiography cannot adequately visualise the left atrial appendage due to its posterior location and acoustic shadowing from lung tissue. This diagnostic limitation means potentially life-threatening thrombi remain hidden from conventional imaging.
The stroke mechanism involves two distinct phases: thrombus formation during prolonged arrhythmia (>24-48 hours), and critically, atrial stunning following cardioversion. When normal rhythm returns, the atria temporarily lose effective contractile function—atrial paresis—creating a paradoxical increase in stroke risk despite rhythm restoration. This phenomenon explains why stroke rates peak in the immediate post-cardioversion period, making pre-procedural thrombus exclusion essential for patient safety.

What the Evidence Shows Multiple large-scale studies demonstrate TOE’s superior diagnostic accuracy for left atrial appendage thrombus detection compared to transthoracic echocardiography. The ACUTE trial showed TOE-guided cardioversion achieved similar stroke rates to prolonged anticoagulation strategies while enabling earlier intervention.
Research reveals that 90% of atrial fibrillation-related thrombi form within the left atrial appendage, with sensitivity of transthoracic echo for detection remaining disappointingly low at 39-63%. In contrast, TOE achieves 95-100% sensitivity for appendage thrombus identification.
The timing of stroke risk proves counterintuitive. While many clinicians focus on pre-existing clots, evidence shows the greatest danger occurs post-cardioversion due to atrial stunning. Studies demonstrate that even successful rhythm conversion can precipitate stroke through this mechanism, with risk persisting for several weeks.
Current NICE guidelines (CG180) recommend either 3-4 weeks of therapeutic anticoagulation before cardioversion, or TOE to exclude thrombus with heparin cover during the procedure. The TOE-guided approach offers particular advantages in symptomatic patients requiring urgent intervention, those with questionable anticoagulation compliance, or cases where rapid rhythm restoration provides significant clinical benefit.
Meta-analyses consistently show stroke rates below 1% when TOE confirms absence of left atrial appendage thrombus, provided appropriate periprocedural anticoagulation protocols are followed.
Clinical Implications TOE before cardioversion transforms patient management by enabling safe, early intervention while maintaining stroke prevention. This approach proves particularly valuable for severely symptomatic patients who cannot tolerate prolonged arrhythmia periods required for traditional anticoagulation strategies.
The procedure requires conscious sedation and specialist expertise, making it unsuitable for emergency department use. However, planned TOE-guided cardioversion often reduces overall hospital admissions by avoiding prolonged pre-treatment periods and repeat presentations with persistent symptoms.
Post-cardioversion anticoagulation remains mandatory regardless of TOE findings, due to atrial stunning risk. Current evidence supports minimum 4-week therapeutic anticoagulation following successful cardioversion, with longer-term decisions based on CHA2DS2-VASc scoring.
Patient selection requires careful consideration of bleeding versus thrombotic risk. TOE carries minimal procedural risks (<0.1% serious complications) but may be contraindicated in patients with significant oesophageal pathology or severe heart failure.
The economic argument for TOE-guided cardioversion strengthens when considering reduced hospital stays, earlier symptom resolution, and improved quality of life compared to prolonged anticoagulation strategies requiring multiple follow-up appointments.
When to Refer Routine referral (within 2-4 weeks):
- AF >24-48 hours duration requiring cardioversion
- Symptomatic AF with rapid ventricular response
- Patients unsuitable for prolonged anticoagulation waiting periods
- Uncertain anticoagulation compliance history
Urgent referral (within 1 week):
- Severe symptoms affecting daily activities
- Heart failure secondary to AF with rapid ventricular response
- Haemodynamic compromise from sustained tachycardia
Emergency referral (same day):
- Haemodynamic instability
- Acute heart failure with AF
- Chest pain with rapid AF
Include in referral: Duration of AF episode, current medications, bleeding risk assessment, recent echo results, CHA2DS2-VASc score, and symptom severity impact on quality of life.
Key Takeaways
- TOE detects 90% of AF-related thrombi in the left atrial appendage that transthoracic echo cannot visualise
- Greatest stroke risk occurs post-cardioversion due to atrial stunning, not pre-existing clots
- TOE-guided cardioversion enables safe early intervention while maintaining stroke prevention
- Four weeks post-cardioversion anticoagulation remains mandatory regardless of TOE findings
Key References
- European Society of Cardiology (ESC) Guidelines for the diagnosis and management of atrial fibrillation
https://www.escardio.org/Guidelines/Clinical-Practice-Guidelines/Atrial-Fibrillation - National Institute for Health and Care Excellence (NICE) Atrial fibrillation: diagnosis and management
https://www.nice.org.uk/guidance/ng196 - American Heart Association / American College of Cardiology / Heart Rhythm Society AF Guideline Update
https://www.ahajournals.org - Manning WJ et al. Left atrial appendage function after cardioversion (atrial stunning)
https://pubmed.ncbi.nlm.nih.gov/ - Cardiac Electrophysiology review on atrial stunning and thromboembolism risk
https://www.ncbi.nlm.nih.gov/
Related Blogs
- Two ways AFib can cause stroke or heart attack
- What is Atrial Fibrillation (AFib)?
- Managing your own Atrial Fibrillation: Taking control of your heart health
- How AFib can present as a heart attack or stroke
Downloads
Printable version